Novel immunotherapy approach designed to stimulate immune response by activating B cells to enhance polyclonal antibody production and potentially reduce hospitalization and progression to respiratory failure
Randomized, double-blind, placebo-controlled study expected to enroll ~1,000 patients across global sites with results expected in Q4 2021
Company to host conference call and webcast today at 8:30 a.m. ET / 5:30 a.m. PT
BURLINGAME, Calif., Feb. 04, 2021 (GLOBE NEWSWIRE) -- Corvus Pharmaceuticals, Inc. (NASDAQ: CRVS), a clinical-stage biopharmaceutical company, today announced that it has initiated a Phase 3 clinical trial of CPI-006 for the treatment of hospitalized patients with COVID-19. The study is expected to enroll approximately 1,000 patients at sites in North America, Europe, South Africa and Latin America.
“The COVID-19 pandemic has been devastating to patients and the healthcare system, and it is critical that we develop new therapies that can improve patient outcomes,” said Gerard J. Criner, MD, FACP, FACCP, Chair and Professor of Thoracic Medicine and Surgery at the Lewis Katz School of Medicine at Temple University and Director of the Temple Lung Center and one of the lead investigators of the trial. “CPI-006 has already shown promising results in COVID-19 patients and its proposed mechanism of stimulating the immune system to specifically target the SARS-CoV-2 virus may have several potential advantages over existing therapies, including the potential to address recently emerging variants of the virus. We look forward to offering this therapy to our patients as part of the Phase 3 study.”
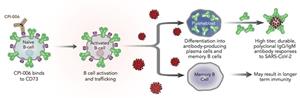
CPI-006 is a humanized monoclonal antibody that is designed to bind to and activate B cells that Corvus believes has the potential to provide a unique immunotherapy approach for the treatment of infectious diseases, including COVID-19. In a Phase 1 study involving 28 hospitalized, high-risk patients with moderate COVID-19 treated with CPI-006, no patients progressed to requiring mechanical ventilation and the median time to discharge from the hospital was 3.5 days. This compares favorably to published reports showing that, on average, approximately 20% of similarly affected patients will progress to requiring invasive mechanical ventilation. Patients in the study generated high titers of polyclonal antibodies against a diverse range of targets on the SARS-CoV-2 virus that were sustained over several months. They also had increased levels of circulating memory B cells, which could lead to long-term immunity.
Serum from 2 of 2 patients treated with CPI-006 early in the pandemic (July 2020), and before variants were discovered, were tested in neutralization assays against both the UK variant and wild type receptor binding domain of SARS-CoV-2 (N501Y mutation). Day 28 post CPI-006 treatment serum showed increases in neutralization titers of 4.5 fold and 1.7 fold to the UK variant for the two patients respectively, compared to the wild type virus. These data will require further confirmation with many more additional patients, but suggest that CPI-006 elicited a broad anti-viral response that may address the problem of immune escape.
The Phase 3 double-blind study, which was designed with guidance from the Food and Drug Administration (FDA), will evaluate the efficacy and safety of CPI-006 compared to placebo in hospitalized patients with mild-to-moderate COVID-19. Patients will be randomized in a 1:1:1 ratio to receive a single intravenous CPI-006 dose of either 2.0 mg/kg or 1.0 mg/kg or placebo; all patients will receive standard of care treatments for COVID-19. The primary endpoint is the proportion of patients progressing to respiratory failure or death during the 28 days after dosing. Respiratory failure is defined as requiring non-invasive or invasive mechanical ventilation. Additional secondary endpoints include time to recovery, time to resolution of COVID-19 symptoms, anti-viral antibody responses, etc. An interim futility and efficacy analysis will be conducted by an independent data monitoring committee when approximately 60% of subjects complete the 28-day post-treatment visit. Results from the study are expected to be available in the fourth quarter 2021.
“The devastating nature of the pandemic and emerging understanding of this disease compels us to rapidly develop CPI-006 for the treatment of COVID-19,” said Richard A. Miller, M.D., president and chief executive officer of Corvus. “We believe CPI-006’s novel mechanism of action, supported by Phase 1 results obtained to date, indicate that this agent may be well suited to combat this virus, which is now demonstrating a propensity to mutate, become more transmissible and potentially escape from current vaccination and therapeutic approaches such as the administration of passive anti-viral monoclonal antibodies. The advancement of this study is the top priority for the Company given the growth in new COVID-19 cases and could provide the foundation for its potential use in other infectious diseases. If successful and ultimately approved for treatment, we believe CPI-006 could be an important therapy if it is able to shorten recovery times and keep COVID-19 patients out of the intensive care unit and off of mechanical ventilators, while also providing durable, longer-term immunity and potential protection against re-infection.”
Corvus also announced that in collaboration with its Chinese partner, Angel Pharmaceuticals, it plans to initiate a global Phase 2 trial with CPI-818, an ITK inhibitor, in refractory peripheral T cell lymphoma by the end of 2021. The Company also announced its plans to collaborate with an academic consortium to evaluate ciforadenant, its A2A receptor antagonist, for the first line treatment of metastatic renal cell carcinoma in combination with pembrolizumab and a tyrosine kinase inhibitor.
Conference Call, Webcast and Slide Presentation Details
Corvus will host a conference call and webcast today, February 4, 2021, at 8:30 a.m. ET (5:30 a.m. PT), to discuss the Phase 3 CPI-006 clinical trial for COVID-19 and other topics. The conference call can be accessed by dialing 1-877-407-0784 (toll-free domestic) or 1-201-689-8560 (international) and using the conference ID 13715838. The live webcast, which will include presentation slides, may be accessed via the investor relations section of the Corvus website. A replay of the webcast will be available on Corvus' website for 90 days.
About Corvus Pharmaceuticals
Corvus Pharmaceuticals is a clinical-stage biopharmaceutical company. Corvus’ lead product candidates are
CPI-006, a humanized monoclonal antibody directed against CD73 that has exhibited immunomodulatory activity and activation of immune cells in preclinical studies, and ciforadenant (CPI-444), a small molecule inhibitor of the A2A receptor. CPI-006 is being evaluated in a Phase 3 clinical trial for the treatment of hospitalized patients with COVID-19 and in a multicenter Phase 1/1b oncology clinical trial as a single agent, in combination with ciforadenant and pembrolizumab. The Company’s third cancer clinical program, CPI-818, is an investigational, oral, small molecule drug that selectively inhibited ITK in preclinical studies, is in a multicenter Phase 1/1b clinical trial in patients with several types of T-cell lymphomas. For more information, visit www.corvuspharma.com.
About CPI-006
CPI-006 is an investigational, potent humanized monoclonal antibody that is designed to react with a specific site on CD73. In preclinical studies, it has demonstrated immunomodulatory activity resulting in activation of lymphocytes, induction of antibody production from B cells and effects on lymphocyte trafficking. While there are other anti-CD73 antibodies and small molecules in development for treatment of cancer, such agents react with a different region of CD73 and are designed to block production of adenosine, which is not involved in the immunomodulatory processes seen with CPI-006.
About CPI-818
CPI-818 is an investigational small molecule drug given orally that has selectively inhibited ITK (interleukin-2-inducible T-cell kinase) in preclinical studies. It was designed to possess dual properties: to block malignant T-cell growth and to modulate immune responses. ITK, an enzyme, is expressed predominantly in T-cells and plays a role in T-cell and natural killer (NK) cell lymphomas and leukemias, as well as in normal immune function. Interference with ITK signaling can modulate immune responses to various antigens. The Company believes the inhibition of specific molecular targets in T-cells may be of therapeutic benefit for patients with T-cell lymphomas and in patients with autoimmune diseases. The Company is conducting a Phase 1/1b trial in patients with refractory T-cell lymphomas.
About Ciforadenant
Ciforadenant (CPI-444) is an investigational small molecule, oral, checkpoint inhibitor designed to disable a tumor’s ability to subvert attack by the immune system by blocking the binding of adenosine in the tumor microenvironment to the A2A receptor. Adenosine, a metabolite of ATP (adenosine tri-phosphate), is produced within the tumor microenvironment where it may bind to the adenosine A2A receptor present on immune cells and block their activity. CD39 and CD73 are enzymes on the surface of tumor cells and immune cells. These enzymes work in concert to convert ATP to adenosine.
About Angel Pharmaceuticals
Angel Pharmaceuticals is a privately held biopharmaceutical company developing a pipeline of precisely targeted investigational medicines for cancer, autoimmune, infectious and other serious diseases in China. Angel Pharmaceuticals was launched through a collaboration with U.S.-based Corvus Pharmaceuticals and investments from investors in China. Angel Pharmaceuticals licensed the rights to develop and commercialize Corvus’ three clinical-stage candidates – ciforadenant, CPI-006 and CPI-818 – in greater China and obtained global rights to Corvus’ BTK inhibitor preclinical programs. Under the collaboration, Corvus initially retained a 49.7% equity stake in Angel Pharmaceuticals and designated three individuals on Angel’s five-person Board of Directors.
One of the main strategic rationales for Corvus’ collaboration with Angel is to gain clinical study synergies and accelerate development timelines, whereby data from patients enrolled in China studies could potentially be used as part of U.S. regulatory submissions as part of a global pivotal study protocol. Based on the tumor responses observed in the CPI-818 Phase 1/1b clinical trial, the companies are planning to collaborate on the next clinical study for CPI-818 for the treatment of T cell lymphomas.
Forward-Looking Statements
This press release contains forward-looking statements, including statements related to the potential safety and efficacy of CPI-006, CPI-818 and ciforadenant, the Company’s ability to develop and advance product candidates into and successfully complete preclinical studies and clinical trials, including the Company’s Phase 3 clinical trial of CPI-006 for COVID-19, the timing of the availability and announcement of clinical data and certain other product development milestones, and the sufficiency of the Company’s cash resources. All statements other than statements of historical fact contained in this press release are forward-looking statements. These statements often include words such as “believe,” “expect,” “anticipate,” “intend,” “plan,” “estimate,” “seek,” “will,” “may” or similar expressions. Forward-looking statements are subject to a number of risks and uncertainties, many of which involve factors or circumstances that are beyond the Company’s control. The Company’s actual results could differ materially from those stated or implied in forward-looking statements due to a number of factors, including but not limited to, risks detailed in the Company’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2020, filed with the Securities and Exchange Commission on October 29, 2020, as well as other documents that may be filed by the Company from time to time with the Securities and Exchange Commission. In particular, the following factors, among others, could cause results to differ materially from those expressed or implied by such forward-looking statements: the Company’s ability to demonstrate sufficient evidence of efficacy and safety in its clinical trials of CPI-006, CPI-818 and ciforadenant; the accuracy of the Company’s estimates relating to its ability to initiate and/or complete preclinical studies and clinical trials; the results of preclinical studies may not be predictive of future results; the unpredictability of the regulatory process; regulatory developments in the United States, and other foreign countries; whether the FDA accepts data from trials conducted in foreign locations, including China; the unpredictability of any ongoing or future trade dispute between the United States and China; the costs of clinical trials may exceed expectations; the Company’s ability to raise additional capital; the effects of COVID-19 on the Company’s clinical programs and business operations. Although the Company believes that the expectations reflected in the forward-looking statements are reasonable, it cannot guarantee that the events and circumstances reflected in the forward-looking statements will be achieved or occur, and the timing of events and circumstances and actual results could differ materially from those projected in the forward-looking statements. Accordingly, you should not place undue reliance on these forward-looking statements. All such statements speak only as of the date made, and the Company undertakes no obligation to update or revise publicly any forward-looking statements, whether as a result of new information, future events or otherwise.
INVESTOR CONTACT:
Leiv Lea
Chief Financial Officer
Corvus Pharmaceuticals, Inc.
+1-650-900-4522
llea@corvuspharma.com
MEDIA CONTACT:
Sheryl Seapy
W2O pure
+1-949-903-4750
sseapy@purecommunications.com
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/4418f37c-2b88-4eb5-95b4-e24383051a5e.

Figure 1
Phase 1 study results, along with pre-clinical research, shows that CPI-006 activates antigen specific B cells, triggering the body’s inherent immune response to generate a robust and durable humoral response to the SARS-CoV-2 virus. This includes stimulating the generation of high titers of durable, polyclonal IgG and IgM antibody responses to SARS-CoV-2 and increased levels of memory B cells. This novel mechanism of action has several advantages over passively administered monoclonal antibodies and could potentially be used to treat other infectious diseases.
Source: Corvus Pharmaceuticals, Inc.