Corvus Pharmaceuticals Announces Data from Cohorts 1-3 of Placebo-Controlled Phase 1 Clinical Trial of Soquelitinib for Atopic Dermatitis
Data continue to demonstrate favorable safety and efficacy profile, including earlier and deeper responses in cohort 3 compared to cohorts 1-2
Data to be presented in an oral session and poster at the
Company to discuss data today on its first quarter 2025 business update conference call and webcast at
“We continue to be encouraged by the results from our Phase 1 trial of soquelitinib in patients with atopic dermatitis, which show a favorable safety and efficacy profile with a convenient oral tablet,” said
Soquelitinib Interim Data from the Atopic Dermatitis Phase 1 Clinical Trial
As of
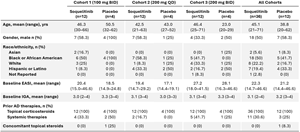
Baseline patient characteristics are shown below in Table 1. Patients enrolled in cohort 3 had more severe disease with higher mean baseline
Table 1: Patient Characteristics
The percent reduction in mean
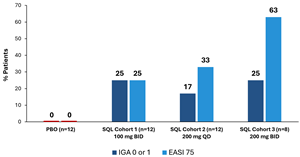
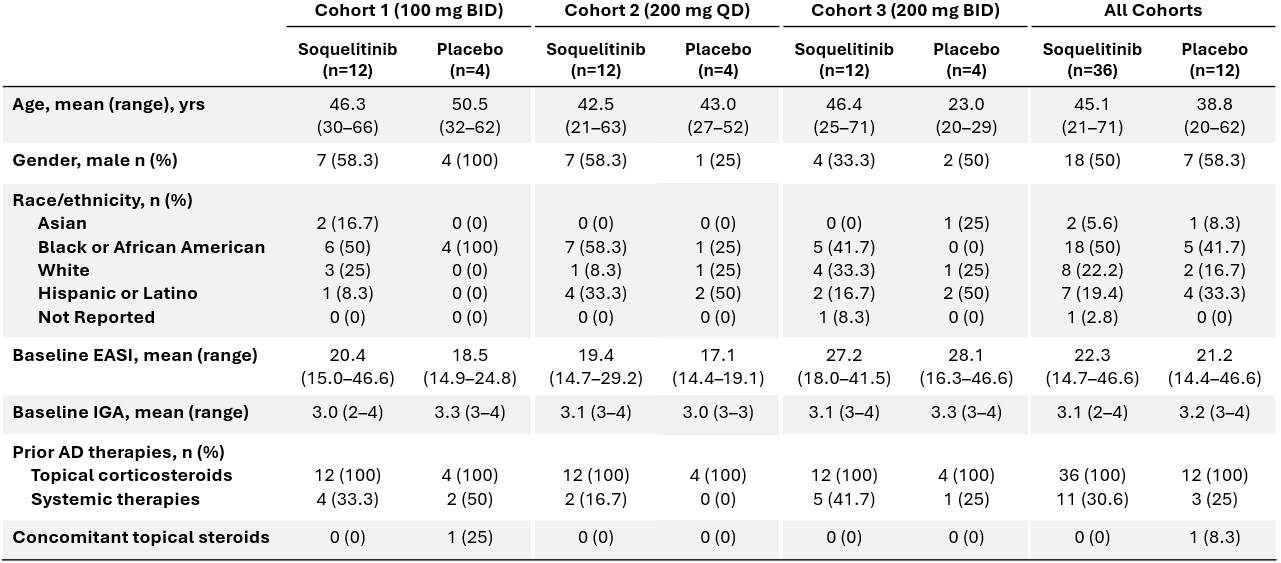
Figure 1 below shows the percent of patients that achieved IGA 0 or 1 or
Figure 1: Percent Patients Achieving Endpoints IGA 0 or 1, EASI 75 at Day 28 of Treatment
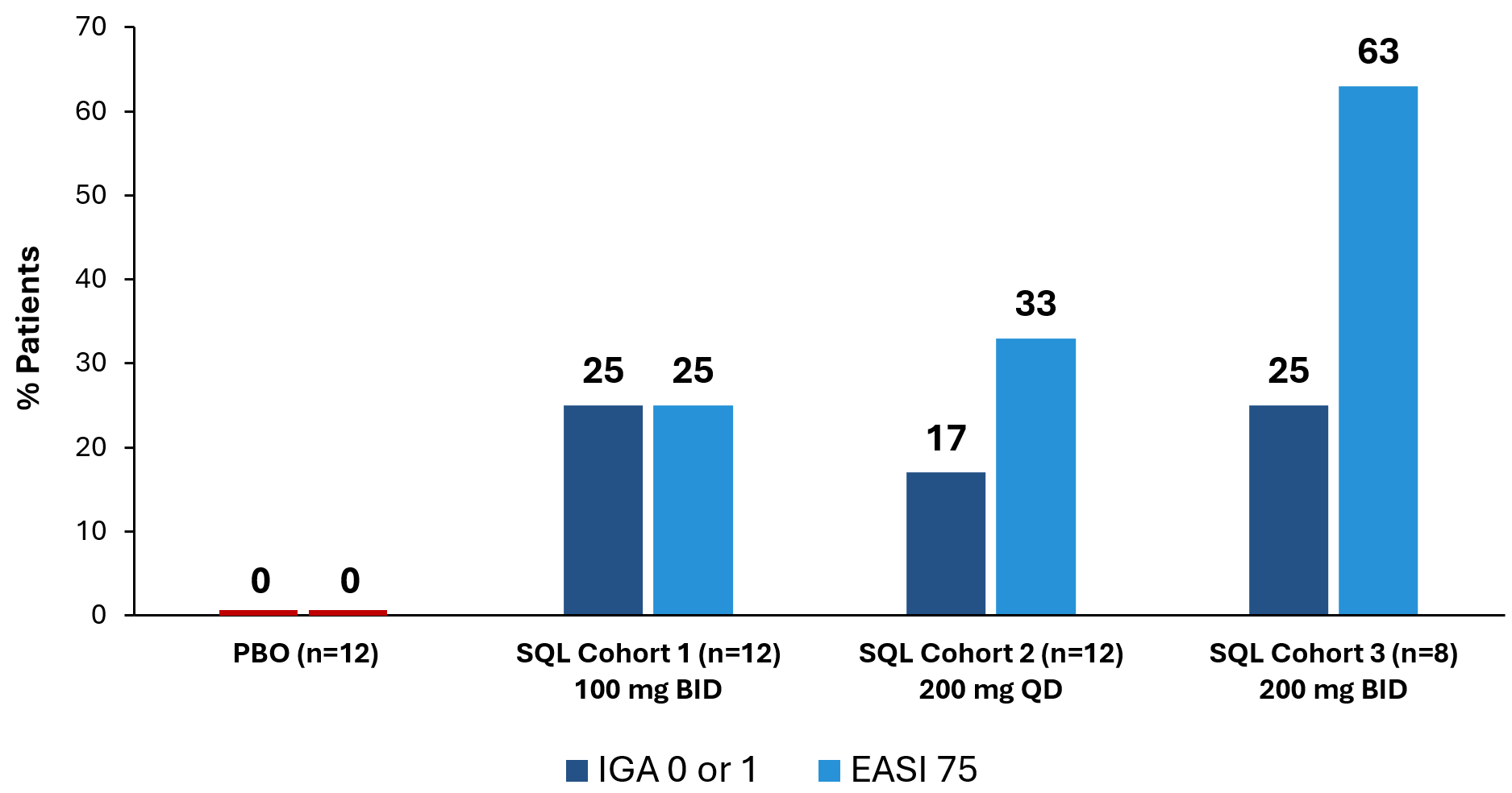
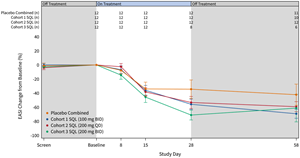
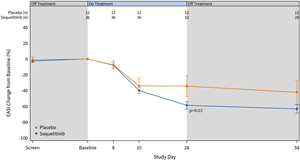
The graphs below (Figures 2 and 3) show the kinetics of response for each of the cohorts and for the combined cohorts 1, 2 and 3. The placebo patients (n=12) are combined in both graphs. Separation of the curves for patients receiving active drug began at day 15 and increased by day 28 for cohorts 1 and 2. Cohort 3 patients experienced earlier and deeper separation from placebo starting by day 8. The combined soquelitinib treatment group is significantly superior to placebo at day 28, p=0.03.
Figure 2: Percent Reduction in Mean EASI Score for Cohorts 1, 2 and 3. Mean percent change in
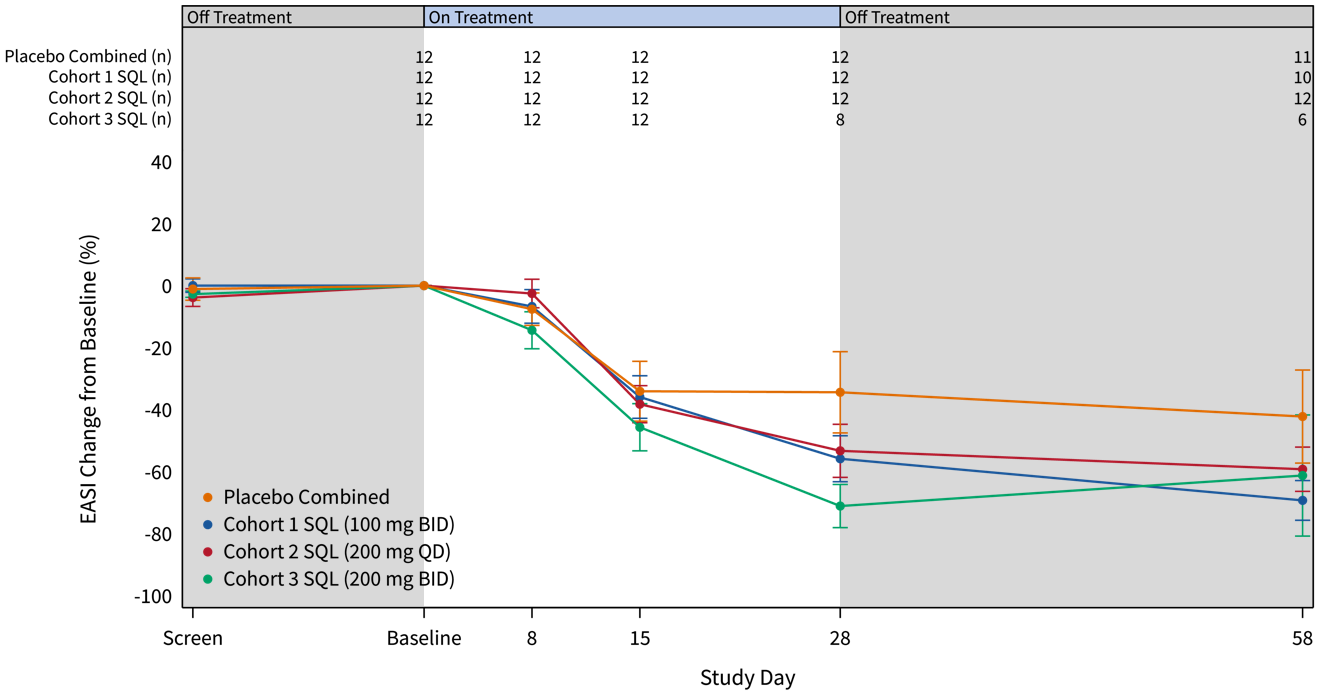
Figure 3: Percent Reduction in Mean EASI Score for Combined Cohorts 1, 2 and 3. The data is displayed below with cohorts combined. Four active patients in cohort 3 have not yet reached day 28 of treatment.
Safety Data
Soquelitinib was well tolerated, with no dose limiting toxicities (DLTs) and no clinically significant laboratory abnormalities observed in any of the cohorts. No interruption of drug dosing was seen in any of the cohorts. Grade 1/2 adverse events (treatment related and unrelated) were seen in 33.3% of patients receiving soquelitinib and 25% receiving placebo. Only one treatment related adverse event of grade 1 nausea was reported with soquelitinib treatment.
Serum Cytokine and Other Biomarker Studies
As reported previously, relationships between reductions in certain cytokines with improvement in
Soquelitinib Atopic Dermatitis Phase 1 Clinical Trial Design and Protocol Update
The randomized, double-blind, placebo-controlled Phase 1 clinical trial was designed to enroll 64 patients with moderate to severe atopic dermatitis that previously failed one prior topical or systemic therapy. Patients were planned to be enrolled into one of four dosing cohorts in a 3:1 ratio (12 active and four placebo) to receive either soquelitinib or placebo. The cohorts are sequentially enrolled and will examine 100 mg orally twice per day, 200 mg orally once per day, 200 mg orally twice per day and 400 mg orally once per day. Patients are treated for 28 days and are then followed for an additional 30 days with no therapy. The Company amended the clinical trial protocol to replace cohort 4 (400 mg once per day) with 24 patients randomized 1:1 between active and placebo. Treatment for this group will be extended to 8 weeks with additional 30-day follow-up with no treatment. The dose level for this group is planned to be the same as cohort 3 – 200 mg orally twice per day.
These doses were selected based on the Company’s prior experience evaluating soquelitinib in T cell lymphoma patients. The doses in the atopic dermatitis trial studied in cohorts 1 and 2 are lower than the 200 mg orally twice a day dosing regimen (same dose as cohort 3 of the atopic dermatitis trial), which is the level that has been shown to provide complete ITK occupancy and that is being evaluated in the Company’s ongoing registrational Phase 3 clinical trial of soquelitinib in peripheral T cell lymphoma.
The primary endpoints include safety and tolerability. Efficacy, measured by improvement in EASI score and IGA, are secondary endpoints. Reduction in itch and various cytokine biomarkers are exploratory endpoints. EASI scores are also evaluated by the percent of patients that achieve a specified percent reduction in EASI score – EASI 50 for patients that achieved a 50% reduction; EASI 75 for a 75% reduction; and EASI 90 for a 90% reduction. Corvus and a data monitoring committee monitor the data from the trial as the trial progresses.
Presentation at
The data from the Phase 1 clinical trial of soquelitinib for atopic dermatitis will be presented by Albert
- Abstract Title: Soquelitinib, a selective ITK inhibitor demonstrates activity in atopic dermatitis phase 1 clinical trial by a novel mechanism of action
- Abstract #: 0437
- Poster Presentation Date and Time:
May 8, 2025 from 4:30 –6:00 pm PT - Oral Presentation Date and Time:
May 10, 2025 from 9:50 – 10:00 am PT
Upcoming Presentation and Webcast
Dr. Miller will present details on the new data from the soquelitinib Phase 1 clinical trial during the Company’s first quarter 2025 business update conference call and webcast today, Thursday, May 8, 2025 at 4:30 pm ET / 1:30 pm PT. The conference call can be accessed by dialing 1-800-717-1738 (toll-free domestic) or 1-646-307-1865 (international) or by clicking on this link for instant telephone access to the event. The live webcast, which will include presentation slides, may be accessed via the investor relations section of the Corvus website. A replay of the webcast will be available on Corvus’ website for 60 days.
About Corvus Pharmaceuticals
Corvus Pharmaceuticals is a clinical-stage biopharmaceutical company pioneering the development of ITK inhibition as a new approach to immunotherapy for a broad range of cancer and immune diseases. The Company’s lead product candidate is soquelitinib, an investigational, oral, small molecule drug that selectively inhibits ITK. Its other clinical-stage candidates are being developed for a variety of cancer indications. For more information, visit www.corvuspharma.com or follow the Company on LinkedIn.
Forward-Looking Statements
This press release contains forward-looking statements related to the potential of the Company’s product candidates including soquelitinib and the interim results from the Phase 1 trial of soquelitinib in patients with atopic dermatitis, the design and timing of initiation of a Phase 2 trial, the advancement of ITK inhibition and the opportunities it provides, and continued advancement of the Company’s clinical pipeline. All statements other than statements of historical fact contained in this press release are forward-looking statements. These statements often include words such as “believe,” “expect,” “anticipate,” “intend,” “plan,” “estimate,” “seek,” “will,” “may” or similar expressions. Forward-looking statements are subject to a number of risks and uncertainties, many of which involve factors or circumstances that are beyond the Company’s control. The Company’s actual results could differ materially from those stated or implied in forward-looking statements due to a number of factors, including but not limited to, risks detailed in the Quarterly Report on Form 10-Q for the first quarter ended
INVESTOR CONTACT:
Chief Financial Officer
+1-650-900-4522
llea@corvuspharma.com
MEDIA CONTACT:
Real Chemistry
+1-949-903-4750
sseapy@realchemistry.com
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/08905c00-1401-44f7-a727-57f812163f24
https://www.globenewswire.com/NewsRoom/AttachmentNg/f00ab519-b9f4-45cd-9a86-2164be1c8931
https://www.globenewswire.com/NewsRoom/AttachmentNg/d0831187-c4ea-4439-b2d1-92922ebedd96
https://www.globenewswire.com/NewsRoom/AttachmentNg/df64aeaa-33f0-4acd-a159-da50d7fc7de9

Source: Corvus Pharmaceuticals, Inc.